May ELISA tests replace IIF in the diagnostic work-up of patients with suspected ANCA-associated vasculitis? The answer is clearly yes according to recently published results of the European Vasculitis Study Group.1
Tag: autoantibodies
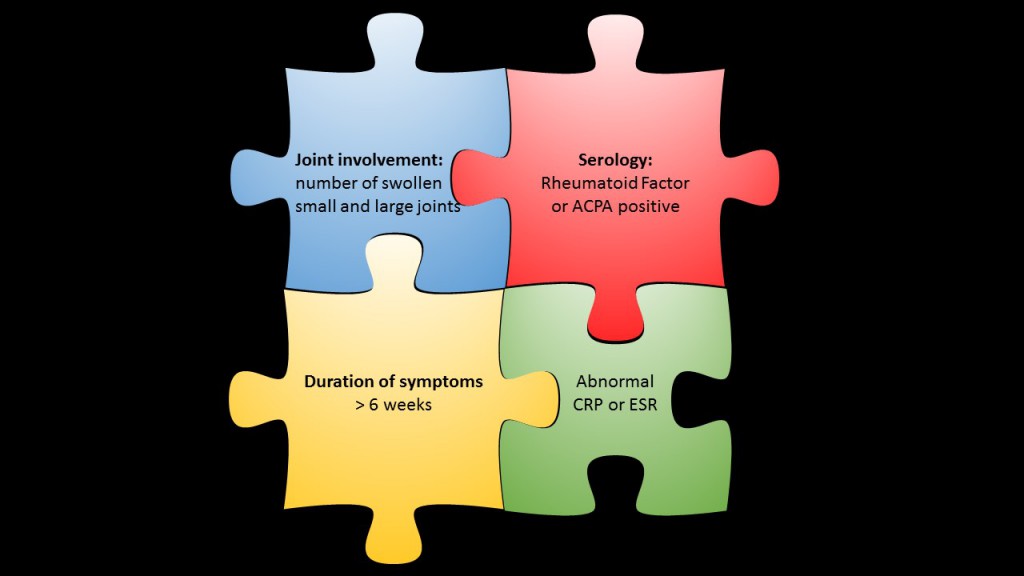
Cutting-edge research for biomarker discovery in rheumatoid arthritis
Towards Early diagnosis and biomarker validation in Arthritis Management
EuroTEAM Arthritis (Towards Early diagnosis and biomarker validation in Arthritis Management) is a challenging research project, funded by the European Union with 5.77 Million Euro for four years. Clinicians and lab scientists with world class expertise in rheumatoid arthritis research from 13 renowned European research institutions and three industrial partners with competence in design and development of diagnostic test kits for autoimmune diseases, local gene therapy for rheumatic diseases, and human genome analysis join their efforts in the discovery of novel biomarkers for early detection of rheumatoid arthritis. The EuroTEAM members intend to develop approaches to predict the onset of rheumatoid arthritis in people who do not yet have the disease. Ultimately, this will help in the development of treatments to prevent people from getting rheumatoid arthritis.
Rheumatoid Factor revisited: An “old” test but still up to date
Rheumatoid factor (RF) is one of the best known serological markers in rheumatology – development of the test dates back into the 1940ies. Since this time the toolkit of serological diagnostic tests for rheumatoid arthritis (RA) has been complemented by the more specific anti-citrullinated protein antibody (ACPA) tests. However, none of the various ACPA tests has completly replaced RF until now.
In contrast, the significance of RF has been further substantiated with the definition of the 2010 ACR criteria for classification of RA. Moreover, recent studies have shown the potential of RF as a contributor to disease pathogenesis.
(more…)
The 2012 revised SLICC criteria for classification of systemic lupus erythematosus

The famous musician Seal is known for his numerous international hits, and for living with an autoimmune disease: the scars on his face are the result of discoid lupus erythematosus. Picture: C. Grube for Access2music.de, wikimedia
Systemic lupus erythematosus (SLE) is a chronic inflammatory disease with manifold manifestations. SLE belongs to the family of autoimmune disorders, diseases that occur, when a mislead immune system attacks the body’s own structures. SLE can affect almost any organ system, thus its presentation and course are highly variable, and diagnosis and therapy may be challenging.
With the intention to classify SLE patients for research and surveillance studies and to support clinicians in confirming a diagnosis, a set of clinical and laboratory classification criteria has been developed and released by the American College of Rheumatology (ACR). The first classification criteria for SLE were originally published in 1971 [1,2]. They have been updated 1982 [3] and 1997 [4] to incorporate new immunologic knowledge and improve patient classification. In contrast to the 1987 criteria, the 1997 criteria have not been validated.
The most recent addendum to the classification criteria for SLE dates from 2012, when the Systemic Lupus International Collaborating Clinics (SLICC) group published a revision and validation of the ACR criteria [5].
Research Update: Rheumatic Disease and Pregnancy
![]() Inflammatory rheumatic diseases predominantly affect women. This also includes many young women who would like to have children or who have not yet completed their family planning when they are first diagnosed. These women do not need to give up on their desire to have children forever. Women with rheumatic disease tend to have fewer children than other women, and it often takes them longer to achieve a desired pregnancy. Today, carefully monitored medical treatment and close collaboration between the rheumatologist and the gynaecologist give these women the opportunity to bring a healthy child into the world.
Inflammatory rheumatic diseases predominantly affect women. This also includes many young women who would like to have children or who have not yet completed their family planning when they are first diagnosed. These women do not need to give up on their desire to have children forever. Women with rheumatic disease tend to have fewer children than other women, and it often takes them longer to achieve a desired pregnancy. Today, carefully monitored medical treatment and close collaboration between the rheumatologist and the gynaecologist give these women the opportunity to bring a healthy child into the world.
Calprotectin for discriminiation of functional and organic bowel disease
 Differential diagnosis of bowel diseases can be challenging, because most of them present with similar common symptoms: abdominal pain and discomfort, diarrhea, weight loss. Infections by common gastrointestinal pathogens may soon be identified, but discrimination of inflammatory bowel disease, of which ulcerative colitis and Crohn’s disease are the most common, and irritable bowel syndrome, remains difficult.
Differential diagnosis of bowel diseases can be challenging, because most of them present with similar common symptoms: abdominal pain and discomfort, diarrhea, weight loss. Infections by common gastrointestinal pathogens may soon be identified, but discrimination of inflammatory bowel disease, of which ulcerative colitis and Crohn’s disease are the most common, and irritable bowel syndrome, remains difficult.
Inflammatory bowel disease (IBD) is characterised by inflammation of the bowel, which is not seen in most patients with irritable bowel syndrome (IBS), and both conditions request different diagnostic and therapeutic approaches. IBD are serious diseases with severe comorbidities, and affected patients need further investigation with extensive diagnostic measures and intensive medical treatment. In contrast to that, IBS may be painful and impairs quality of life, but it does not usually cause serious morbidity. However, patients with IBS can have symptoms for many years and they often experience unnecessary and stressing diagnostic procedures.
Portrait of the HEp2 cell, the pet of immunofluorescence professionals
HEp2 cells are held dear in autoimmune diagnostics. They are invaluable for people engaged in analysing autoantibodies, as E. coli is for molecular biologists or mice for toxicologists.
In spite of a wide range of other suitable methods and technologies, determination of autoantibodies with indirect immuno-fluorescence assays (IFA) on human epithelioma (HEp2) cells still contributes significantly to the diagnosis of autoimmune diseases. The widely recognised advantages of this method are high sensitivity and a broad spectrum of antibodies that can be analysed simultaneously. In addition to mere detection of antibodies a characteristic fluorescence pattern and staining of metaphase and cytoplasmic cells offer supplementary information.
When an autoimmune disease is suspected, the HEp-2 test usually is the first line test. Any positive result is then followed up by a step-wise diagnostic approach, including other immunological tests like ELISA (enzyme-linked immunosorbent assay) for single antibody specificities or immunoblot tests.
(more…)
Immunofluorescence Tests in Crescentic Glomerulonephritis
The web community GRÜNER CLUB AUTOIMMUN is a voluntary association of scientists, laboratory specialists, medical doctors, students and immunofluorescence enthusiasts from Austria. In their internet blog these experienced IFT professionals discuss questions, ideas and concepts of immunofluorescence tests in autoimmune disease diagnostics.
In a recent posting my Austrian colleague Barbara Fabian, community manager of GRÜNER CLUB AUTOIMMUN, refered to the relationship between the formation of autoantibodies against glomerular basal membrane (GBM) and ANCA (antibodies against cytoplasmic antigens of neutrophil granulocytes). The article was originally written in German language, but we had it translated for the not German speaking readers of the Autoimmunity Blog.
Autoantibodies against Glomerular Basal Membrane and Myeloperoxidase in Crescentic Glomerulonephritis
by Barbara Fabian, Vienna
Crescentic Glomerulonephritis (CGN) is an autoimmune disease of the kidney that leads to vasculitis of the capillaries in the glomeruli. The appearance of characteristic autoantibodies or antibody complexes is indicative of CGN and allows for the differentiation of three groups:
Research Update: Osteoimmunology
Linking autoantibody production to bone loss
in rheumatoid arthritis
Autoantibodies against citrullinated proteins (ACPA) are found in people with rheumatoid arthritis and are one of the strongest risk factors for bone destruction in this disease. A recent study now directly links the formation of antibodies binding to mutated citrullinated vimentin (anti-MCV) to bone loss in rheumatoid arthritis, indicating that these autoantibodies act on osteoclasts, the bone cells responsible for bone resorption.
The research of U. Harre, G. Schett and their coworkers provides fundamental new insights into the interaction between bone and the immune system in the inflammatory process leading to the development of rheumatoid arthritis.
Research Update: New Classification Criteria for Sjoegren’s Syndrome
Since the early 1960s almost a dozen different criteria for Sjögren’s syndrome (SS) have been published, both for classifying and for diagnosing that autoimmune disease. Recently, an international team of rheumatologists has published new classification criteria for Sjögren’s syndrome. In the April issue of the Arthritis Care & Research journal the authors propose clear and carefully worded guidelines.
Without question, these “new 2012 classification criteria for Sjögren’s syndrome” are urgently needed to better support etiologic and genetic research and therapeutic trials for Sjögren’s syndrome. Indeed, the new criteria are the first to be based solely on objective clinical tests!
Many other criterions have permitted various testing subjectivity to enable the classification of the disorder. In consequence, subjectivity has made standardisation of clinical trial inclusion something of a moving target, limiting comparability of research data across studies and impeding the needed robust clinical evaluation of possible new treatments. But criteria used for enrollment into clinical trials need to be clear, be easy to apply. And the new 2012 criteria agree to that demand. (more…)